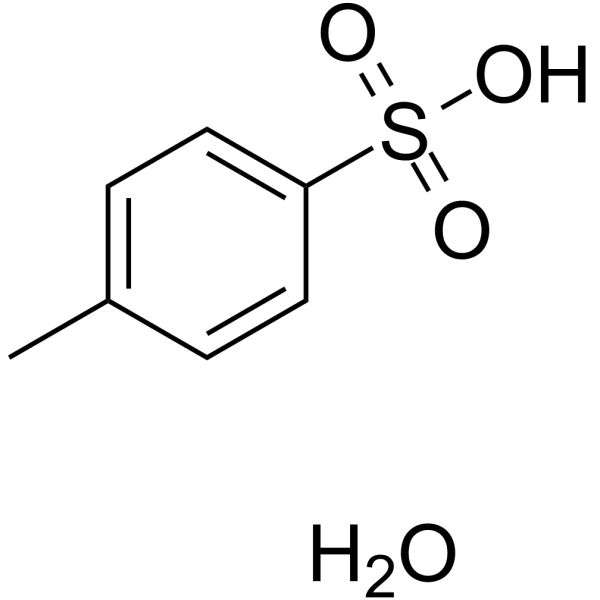
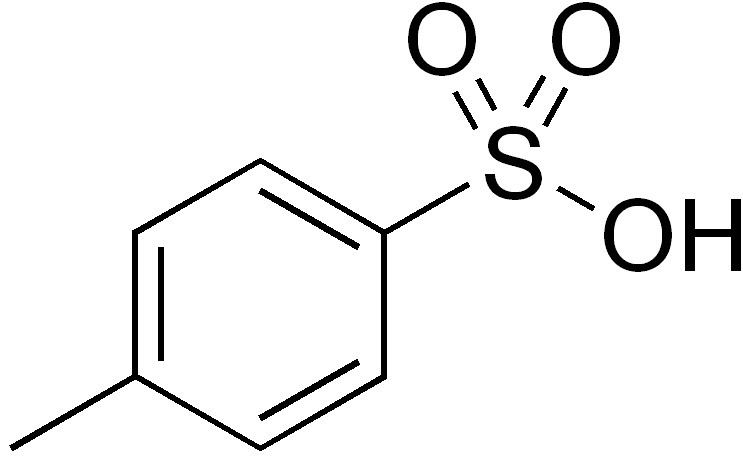
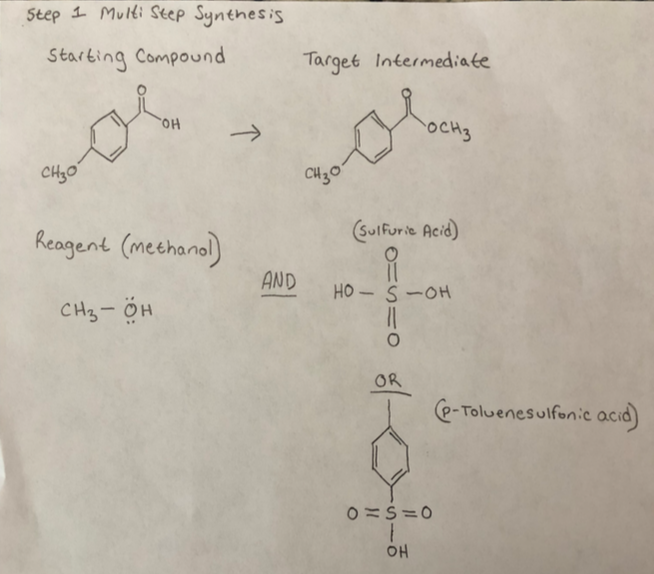
p-Toluenesulfonic acid mediated hydroarylation of cinnamic acids with anisoles and phenols under metal and solvent-free conditions - ScienceDirect

Catalytic systems containing p-toluenesulfonic acid monohydrate catalyzed the synthesis of triazoloquinazolinone and benzimidazoquinazolinone derivatives | SpringerLink
![PDF] p‐Toluenesulfonic Acid Catalyzed Rapid and Efficient Protocol for One‐Pot Synthesis of α‐Amino Nitriles. | Semantic Scholar PDF] p‐Toluenesulfonic Acid Catalyzed Rapid and Efficient Protocol for One‐Pot Synthesis of α‐Amino Nitriles. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/372e6494623b761b605c1716e0e944ea9d76ba69/3-TableI-1.png)
PDF] p‐Toluenesulfonic Acid Catalyzed Rapid and Efficient Protocol for One‐Pot Synthesis of α‐Amino Nitriles. | Semantic Scholar

para-Toluenesulfonic Acid Catalyzed Synthesis of Indenes via a Tandem Friedel–Crafts Alkylation/Hydroarylation of Tertiary Propargylic Alcohols with Electron-Rich Arenes | SpringerLink

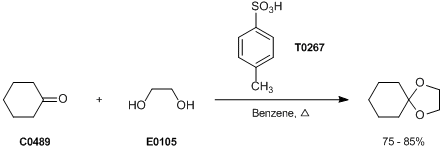
Synthetic Procedures for Compounds 5 and 6 (A) p-Toluenesulfonic acid... | Download Scientific Diagram
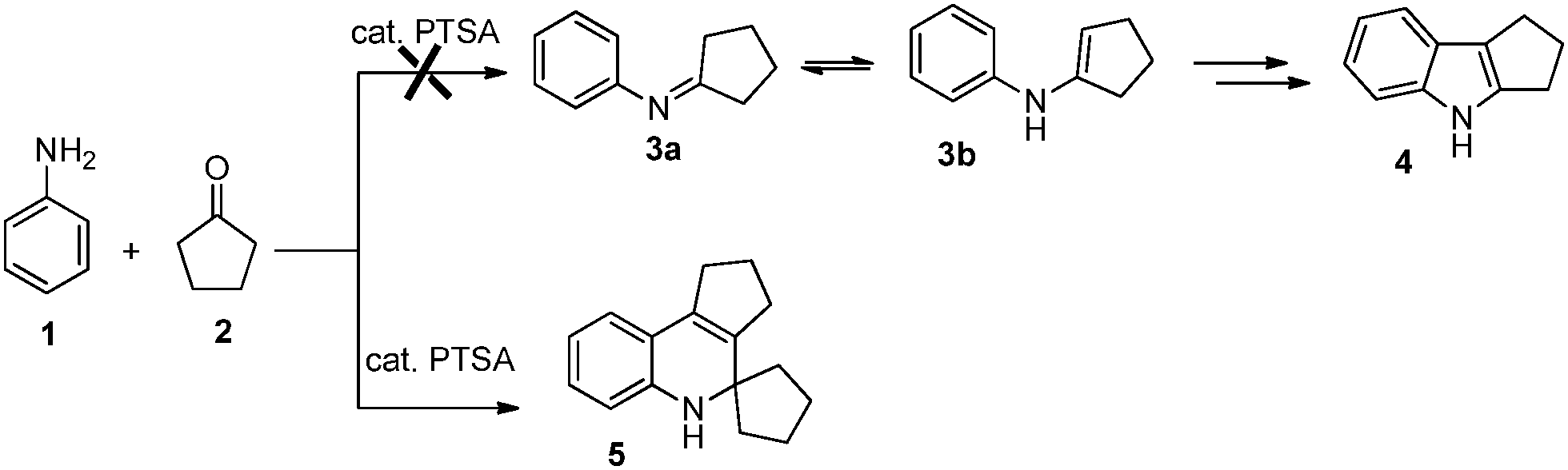
The p -toluenesulfonic acid catalyzed single pot synthesis of tetracyclic 1,2-dihydroquinolines: a metal free approach - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C6NJ00928J

p-Toluene sulfonic acid (PTSA)-MCM-41 as a green, efficient and reusable heterogeneous catalyst for the synthesis of jasminaldehyde under solvent-free condition - ScienceDirect
![p-Toluenesulfonic acid-catalyzed one-pot synthesis of 2-amino-4-substituted-1,4-dihydrobenzo[4,5]imidazolo[1,2-a]pyrimidine-3-carbonitriles under neat conditions - ScienceDirect p-Toluenesulfonic acid-catalyzed one-pot synthesis of 2-amino-4-substituted-1,4-dihydrobenzo[4,5]imidazolo[1,2-a]pyrimidine-3-carbonitriles under neat conditions - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S1631074813002646-sc2.jpg)
p-Toluenesulfonic acid-catalyzed one-pot synthesis of 2-amino-4-substituted-1,4-dihydrobenzo[4,5]imidazolo[1,2-a]pyrimidine-3-carbonitriles under neat conditions - ScienceDirect

Catalytic systems containing p-toluenesulfonic acid monohydrate catalyzed the synthesis of triazoloquinazolinone and benzimidazoquinazolinone derivatives | SpringerLink
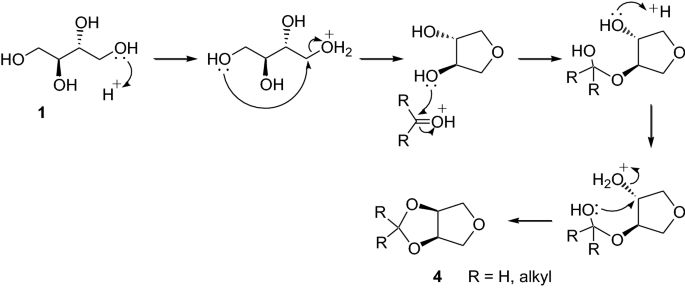
A comparison of homogeneous and heterogeneous Brønsted acid catalysts in the reactions of meso-erythritol with aldehyde/ketones | SpringerLink

Thia‐Michael Addition: An Emerging Strategy in Organic Synthesis - Wadhwa - 2018 - Asian Journal of Organic Chemistry - Wiley Online Library

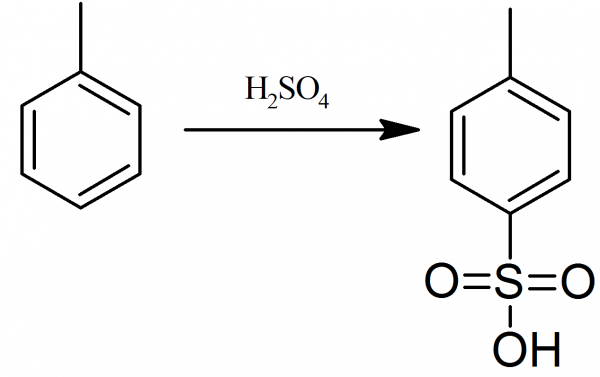
Schematic of the reaction between p-toluenesulfonic acid monohydrate... | Download Scientific Diagram

PDF) One-pot multicomponent synthesis of highly substituted piperidines using p-toluenesulfonic acid monohydrate as catalyst | Nourallah Hazeri - Academia.edu

Schematic of the reaction between p-toluenesulfonic acid monohydrate... | Download Scientific Diagram
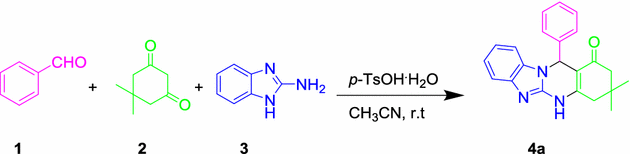
Catalytic systems containing p-toluenesulfonic acid monohydrate catalyzed the synthesis of triazoloquinazolinone and benzimidazoquinazolinone derivatives | SpringerLink

para-Toluenesulfonic Acid Catalyzed Synthesis of Indenes via a Tandem Friedel–Crafts Alkylation/Hydroarylation of Tertiary Propargylic Alcohols with Electron-Rich Arenes | SpringerLink