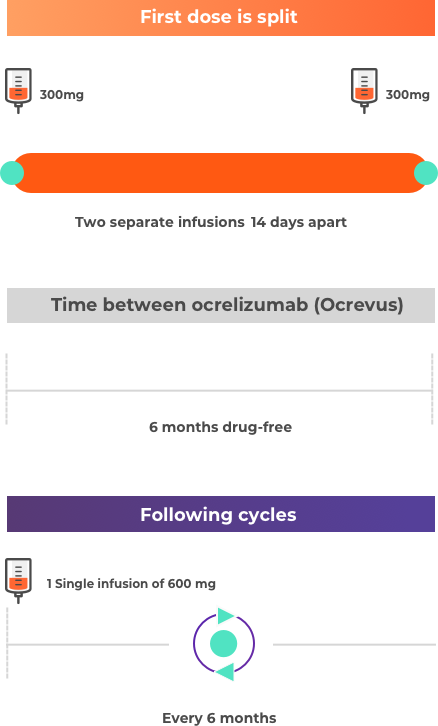
PDF) Shorter infusion time of ocrelizumab: Results from the randomized, double-blind ENSEMBLE PLUS substudy in patients with relapsing-remitting multiple sclerosis
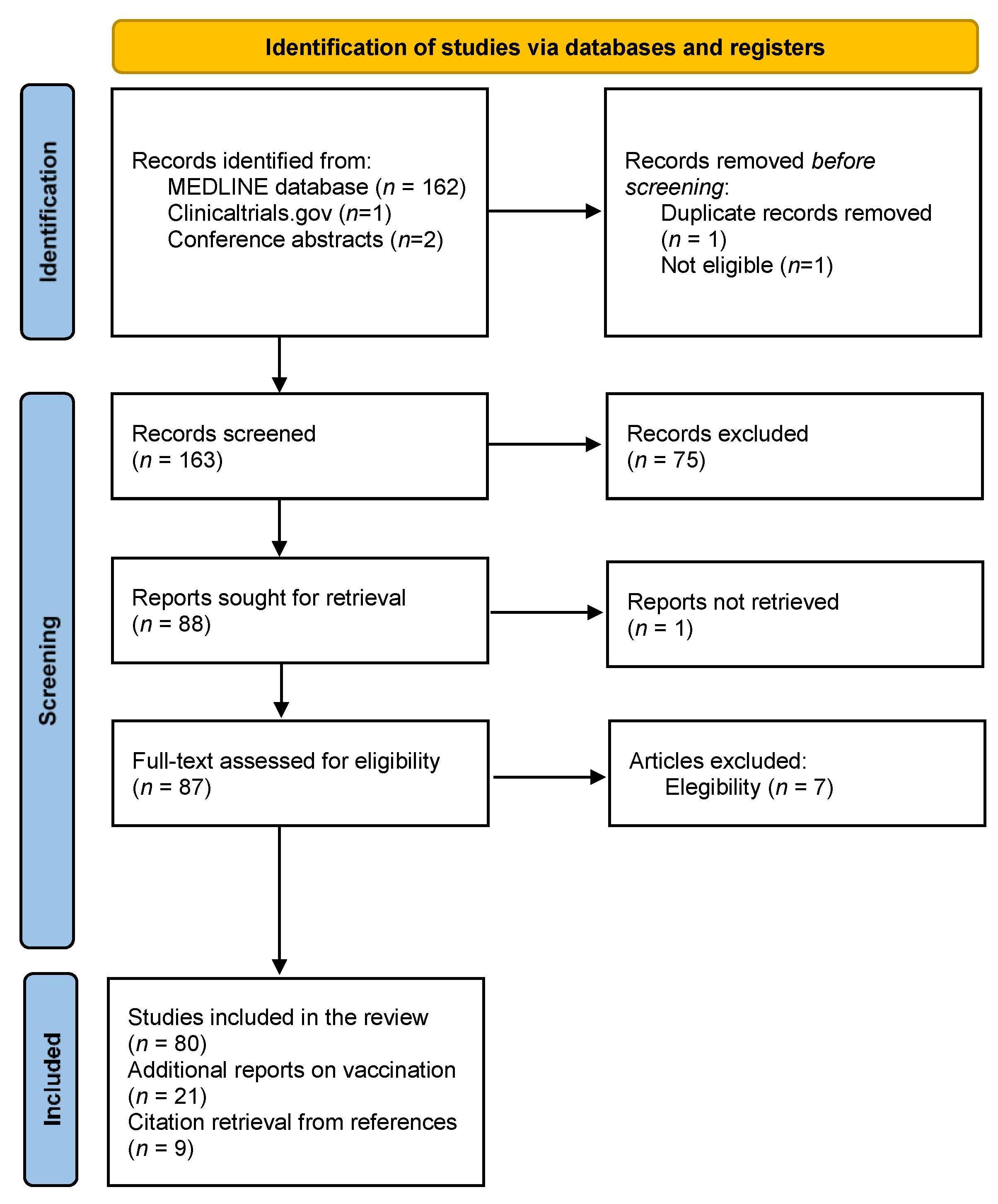
Vaccines | Free Full-Text | Multiple Sclerosis, Disease-Modifying Therapies and COVID-19: A Systematic Review on Immune Response and Vaccination Recommendations | HTML

Een medisch-wetenschappelijk onderzoek om te kijken naar de veiligheid en effectiviteit van een hogere dosis ocrelizumab voor de behandeling van proefpersonen met actieve MS (d.w.z. met terugkomende aanvallen) (Musette)
This is an Extension Study of the Roche P-trial to Investigate Safety and Effectiveness of a Single Ocrelizumab Dose in Participants With Multiple Sclerosis (MS)
![ESCMID Study Group for Infections in Compromised Hosts (ESGICH) Consensus Document on the safety of targeted and biological therapies: an infectious diseases perspective (Agents targeting lymphoid cells surface antigens [I]: CD19, CD20 ESCMID Study Group for Infections in Compromised Hosts (ESGICH) Consensus Document on the safety of targeted and biological therapies: an infectious diseases perspective (Agents targeting lymphoid cells surface antigens [I]: CD19, CD20](https://els-jbs-prod-cdn.jbs.elsevierhealth.com/cms/attachment/71089af2-c3c6-4866-9f65-ae959ab22bf4/gr1_lrg.jpg)
ESCMID Study Group for Infections in Compromised Hosts (ESGICH) Consensus Document on the safety of targeted and biological therapies: an infectious diseases perspective (Agents targeting lymphoid cells surface antigens [I]: CD19, CD20

Efficacy and safety of ocrelizumab in patients with relapsing‐remitting multiple sclerosis with suboptimal response to prior disease‐modifying therapies: A primary analysis from the phase 3b CASTING single‐arm, open‐label trial - Vermersch - -

Efficacy and safety of ocrelizumab in patients with relapsing‐remitting multiple sclerosis with suboptimal response to prior disease‐modifying therapies: A primary analysis from the phase 3b CASTING single‐arm, open‐label trial - Vermersch - -

PDF) Ocrelizumab shorter infusion: Primary results from the ENSEMBLE PLUS substudy in patients with MS

Shorter infusion time of ocrelizumab: Results from the randomized, double-blind ENSEMBLE PLUS substudy in patients with relapsing-remitting multiple sclerosis - Multiple Sclerosis and Related Disorders

Een medisch-wetenschappelijk onderzoek om te kijken naar de veiligheid en effectiviteit van een hogere dosis ocrelizumab voor de behandeling van proefpersonen met actieve MS (d.w.z. met terugkomende aanvallen) (Musette)

PDF) Shorter infusion time of ocrelizumab: Results from the randomized, double-blind ENSEMBLE PLUS substudy in patients with relapsing-remitting multiple sclerosis

Een medisch-wetenschappelijk onderzoek om te kijken naar de veiligheid en effectiviteit van een hogere dosis ocrelizumab voor de behandeling van proefpersonen met actieve MS (d.w.z. met terugkomende aanvallen) (Musette)