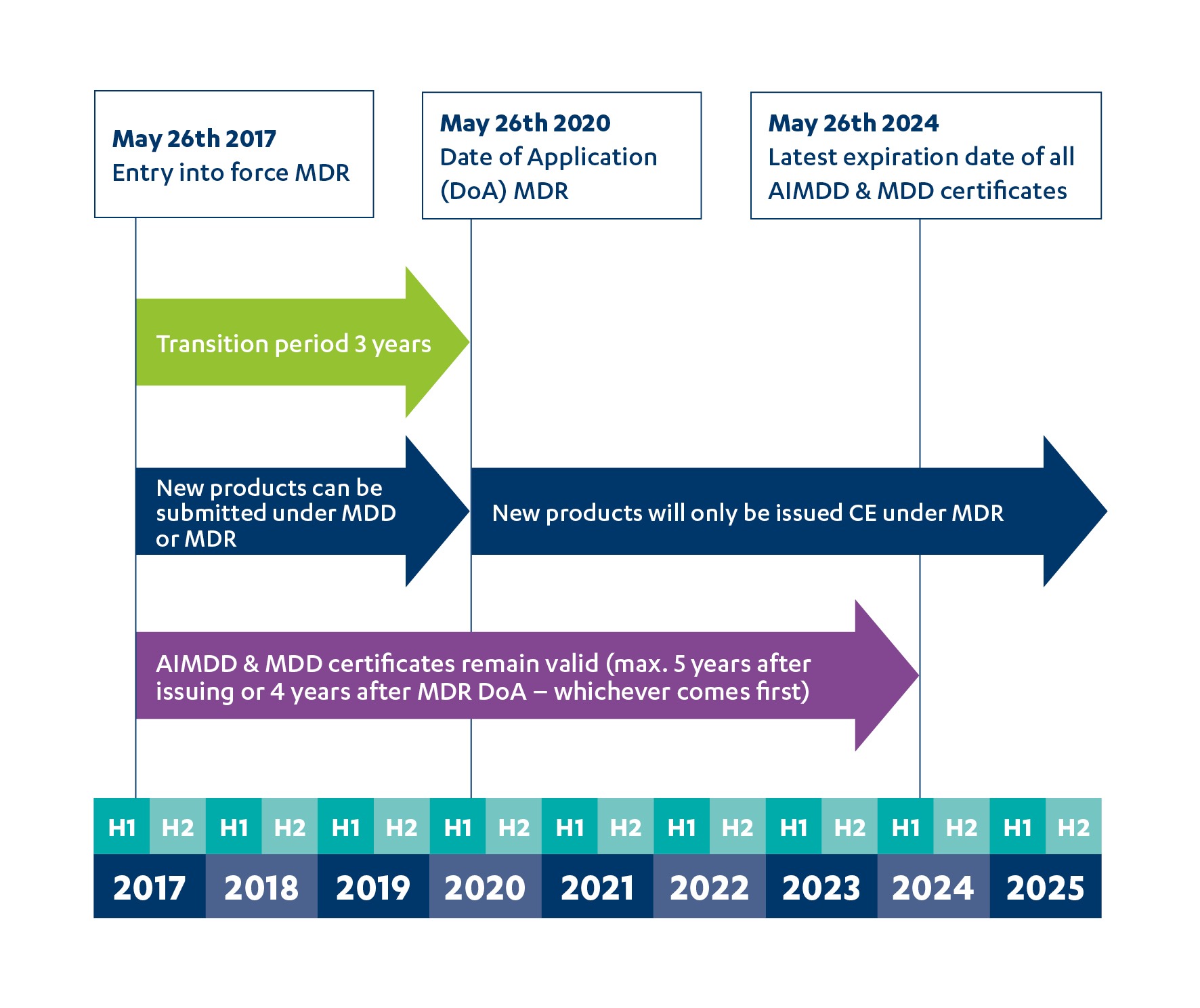
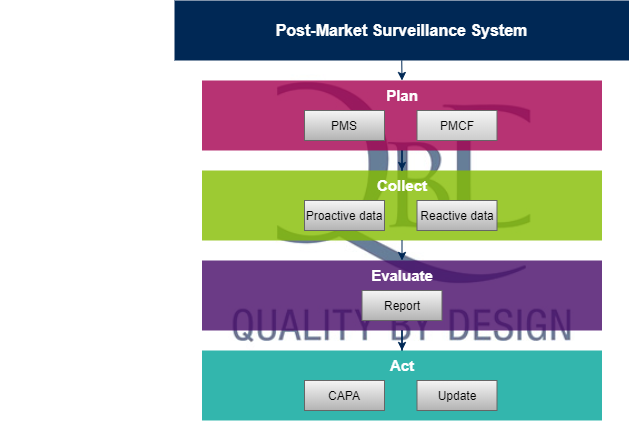
Approval of artificial intelligence and machine learning-based medical devices in the USA and Europe (2015–20): a comparative analysis - The Lancet Digital Health

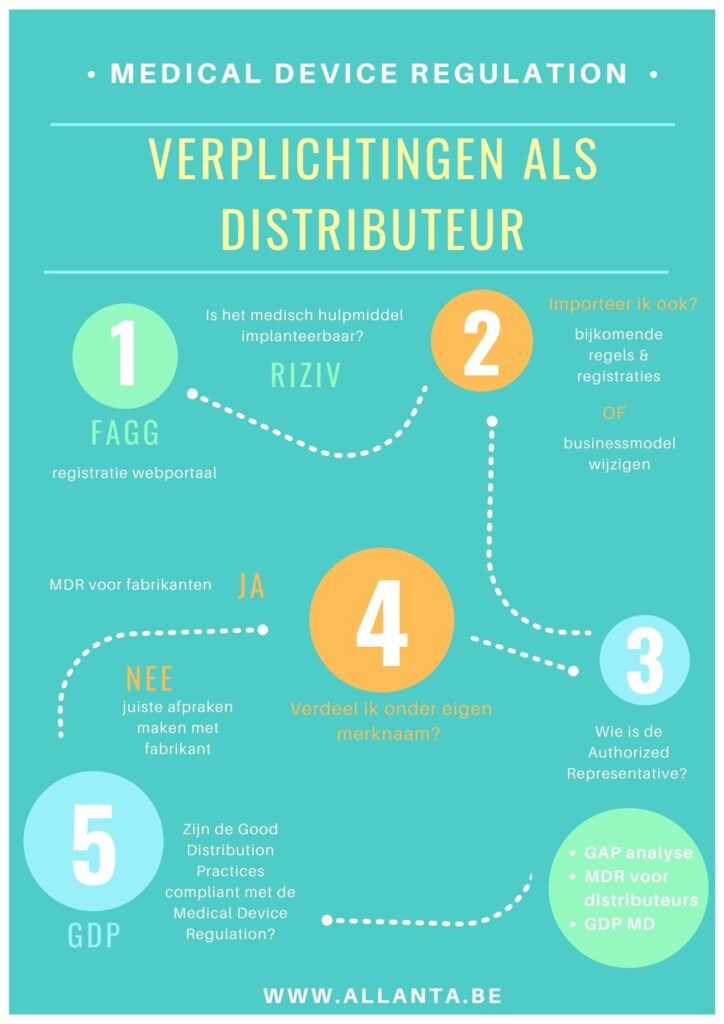
Opleiding MDR - Medical Device Regulation | Eisen voor fabrikanten - Bedrijfsinterne opleiding - Allanta Opleidingen & Advies

Medical device regulation in Europe – what is changing and how can I become more involved? - EuroIntervention