Catalytic Aerobic Oxidation of C(sp3)−H Bonds - Sterckx - 2019 - Angewandte Chemie International Edition - Wiley Online Library

Direct Access to 4-Substituted Isoquinolones via a Sequential Pd-Catalyzed Cyclization/Base-Promoted Aromatization/Ring-Opening of N-Propargyl-1,3-oxazolidines - ScienceDirect

Biosynthesis of UDP-L-Ara4N and transfer of the L-Ara4N unit to lipid... | Download Scientific Diagram

Absence of N-terminal acetyltransferase diversification during evolution of eukaryotic organisms | Scientific Reports
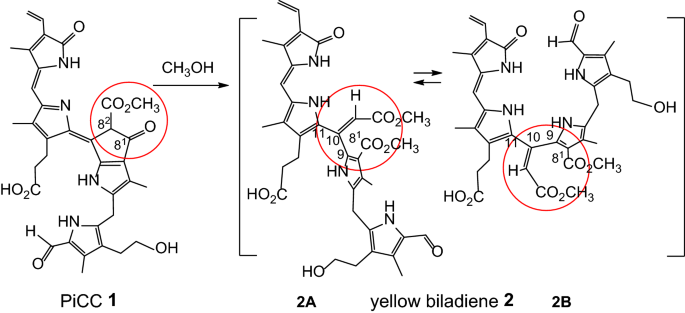
Facile retro-Dieckmann cleavage of a pink phyllobilin: new type of potential downstream steps of natural chlorophyll breakdown | SpringerLink

Direct Access to 4-Substituted Isoquinolones via a Sequential Pd-Catalyzed Cyclization/Base-Promoted Aromatization/Ring-Opening of N-Propargyl-1,3-oxazolidines - ScienceDirect
![Thiazolo[5,4- d ]thiazoles – promising building blocks in the synthesis of semiconductors for plastic electronics - RSC Advances (RSC Publishing) DOI:10.1039/C3RA40851E Thiazolo[5,4- d ]thiazoles – promising building blocks in the synthesis of semiconductors for plastic electronics - RSC Advances (RSC Publishing) DOI:10.1039/C3RA40851E](https://pubs.rsc.org/image/article/2013/RA/c3ra40851e/c3ra40851e-s4.gif)
Thiazolo[5,4- d ]thiazoles – promising building blocks in the synthesis of semiconductors for plastic electronics - RSC Advances (RSC Publishing) DOI:10.1039/C3RA40851E
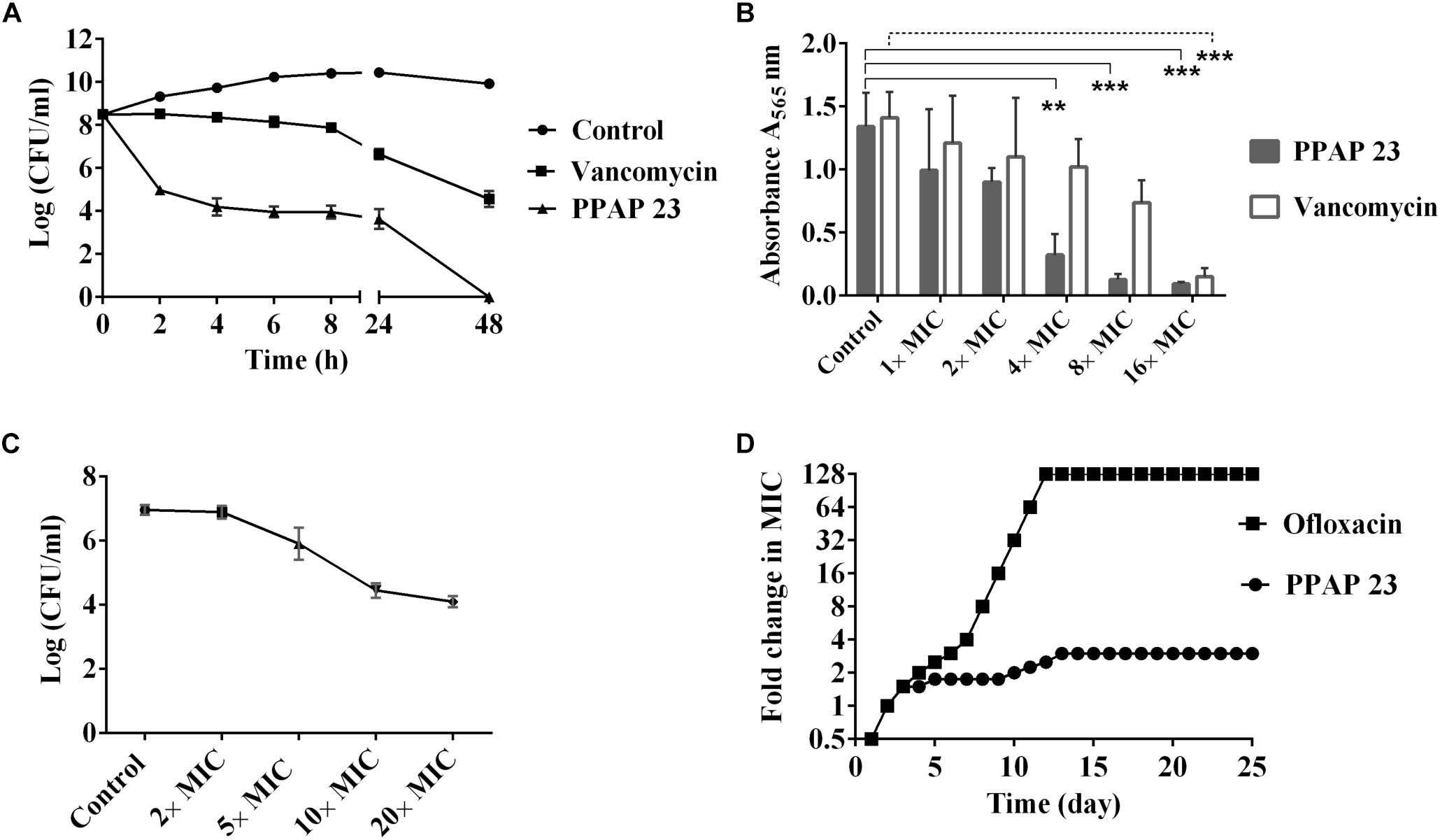
Frontiers | The Polycyclic Polyprenylated Acylphloroglucinol Antibiotic PPAP 23 Targets the Membrane and Iron Metabolism in Staphylococcus aureus | Microbiology

Scheme 1. (a) MMTrCl/Py; (b) TMSCl/Py; (c) BzCl or Ac 2 O or (ClCH 2... | Download Scientific Diagram
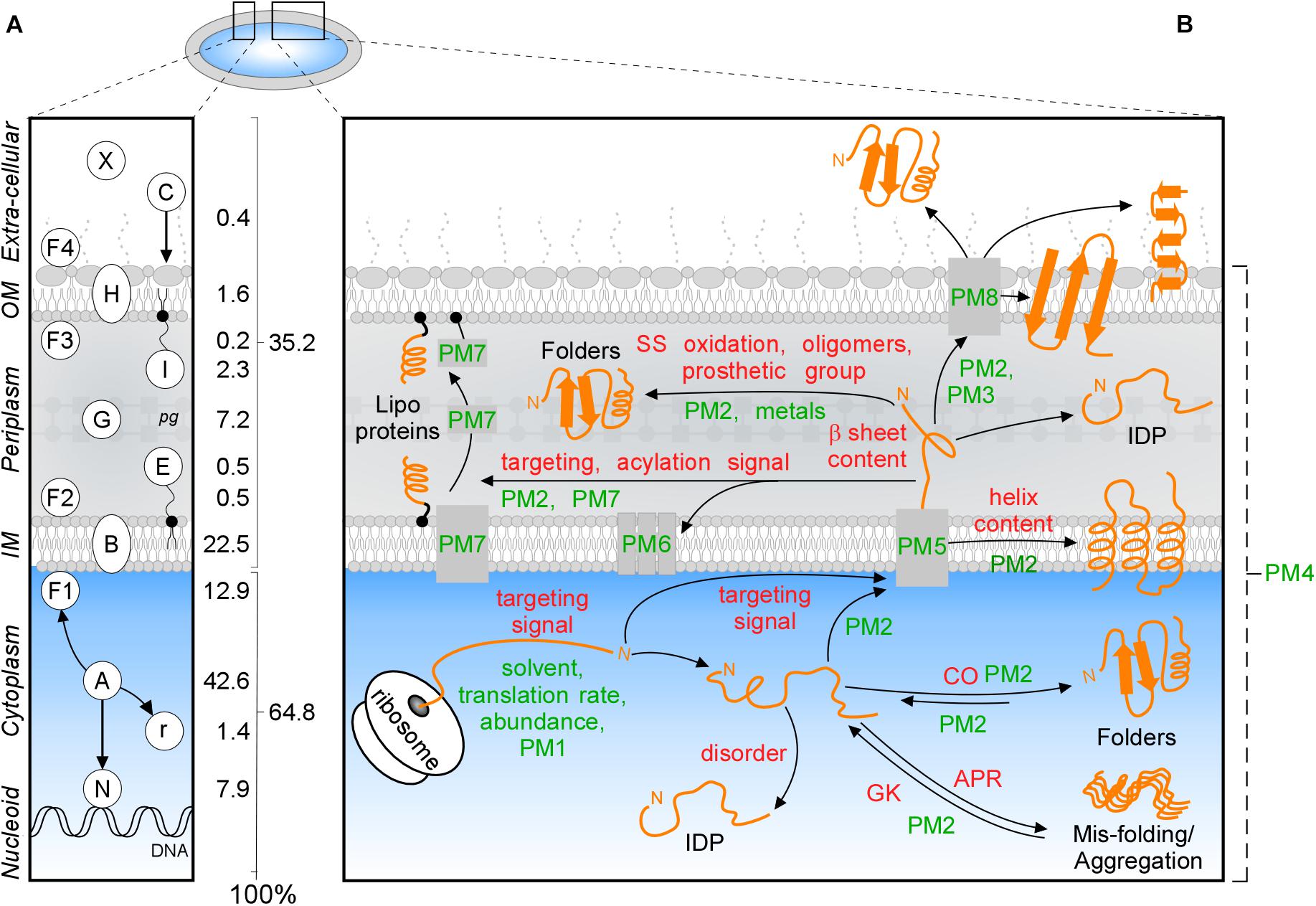
Frontiers | Structural Basis of the Subcellular Topology Landscape of Escherichia coli | Microbiology

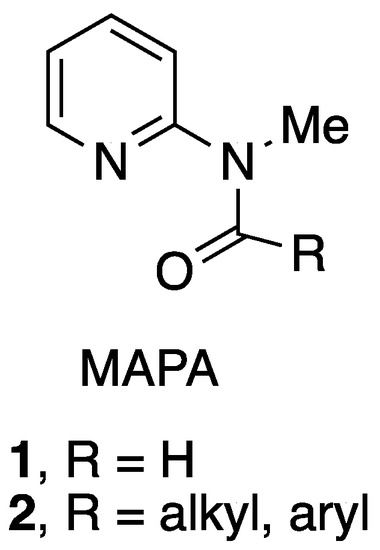
Molecules | Free Full-Text | Synthesis of MAPA Reagents and 2-Alkyl(aryl)aminopyridines from 2-Bromopyridine Using the Goldberg Reaction | HTML
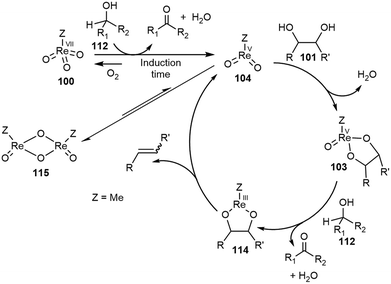
The deoxydehydration (DODH) reaction: a versatile technology for accessing olefins from bio-based polyols - Green Chemistry (RSC Publishing) DOI:10.1039/D0GC00689K
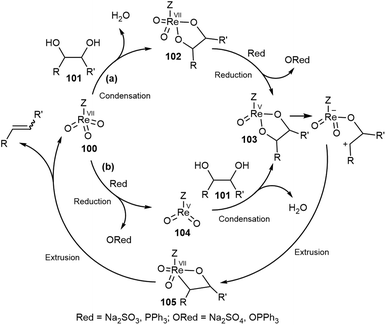
The deoxydehydration (DODH) reaction: a versatile technology for accessing olefins from bio-based polyols - Green Chemistry (RSC Publishing) DOI:10.1039/D0GC00689K

Reaction mechanism of the methylation of a carboxylic acid R-COOH with... | Download Scientific Diagram

Metal‐Free Oxidative CC Bond Formation through CH Bond Functionalization - Narayan - 2015 - Chemistry – A European Journal - Wiley Online Library

Formation of naphthalene hydrates in the enzymatic conversion of 1,2-dihydronaphthalene by two fungal peroxygenases and subsequent naphthalene formation - ScienceDirect

Biosynthesis of UDP-L-Ara4N and transfer of the L-Ara4N unit to lipid... | Download Scientific Diagram
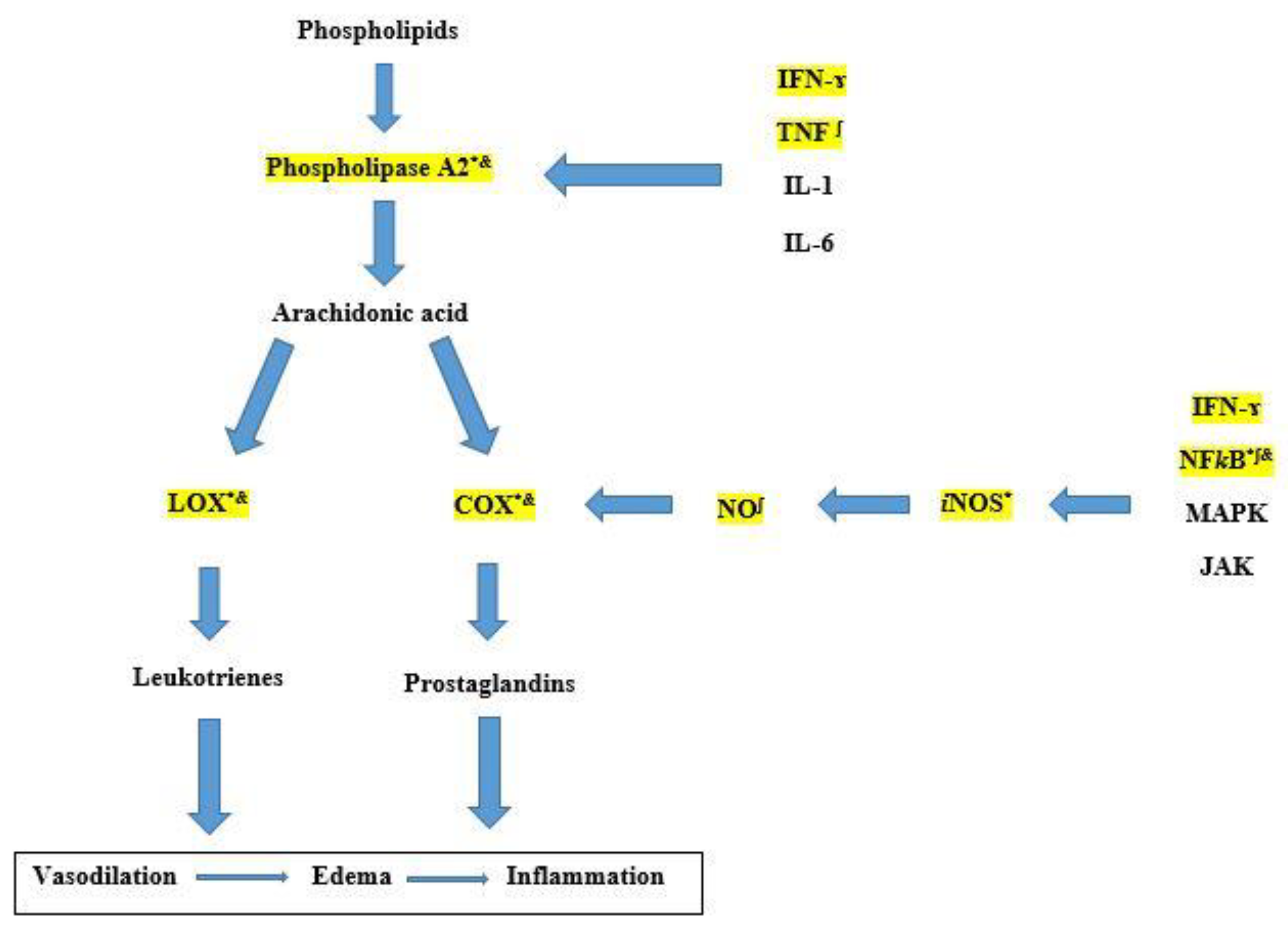
Pharmaceuticals | Free Full-Text | Anti-Inflammatory Potential of Daturaolone from Datura innoxia Mill.: In Silico, In Vitro and In Vivo Studies | HTML

N-terminomics identifies Prli42 as a membrane miniprotein conserved in Firmicutes and critical for stressosome activation in Listeria monocytogenes | Nature Microbiology

Possible reaction mechanism of the hydrolysis of S -formylglutathione... | Download Scientific Diagram